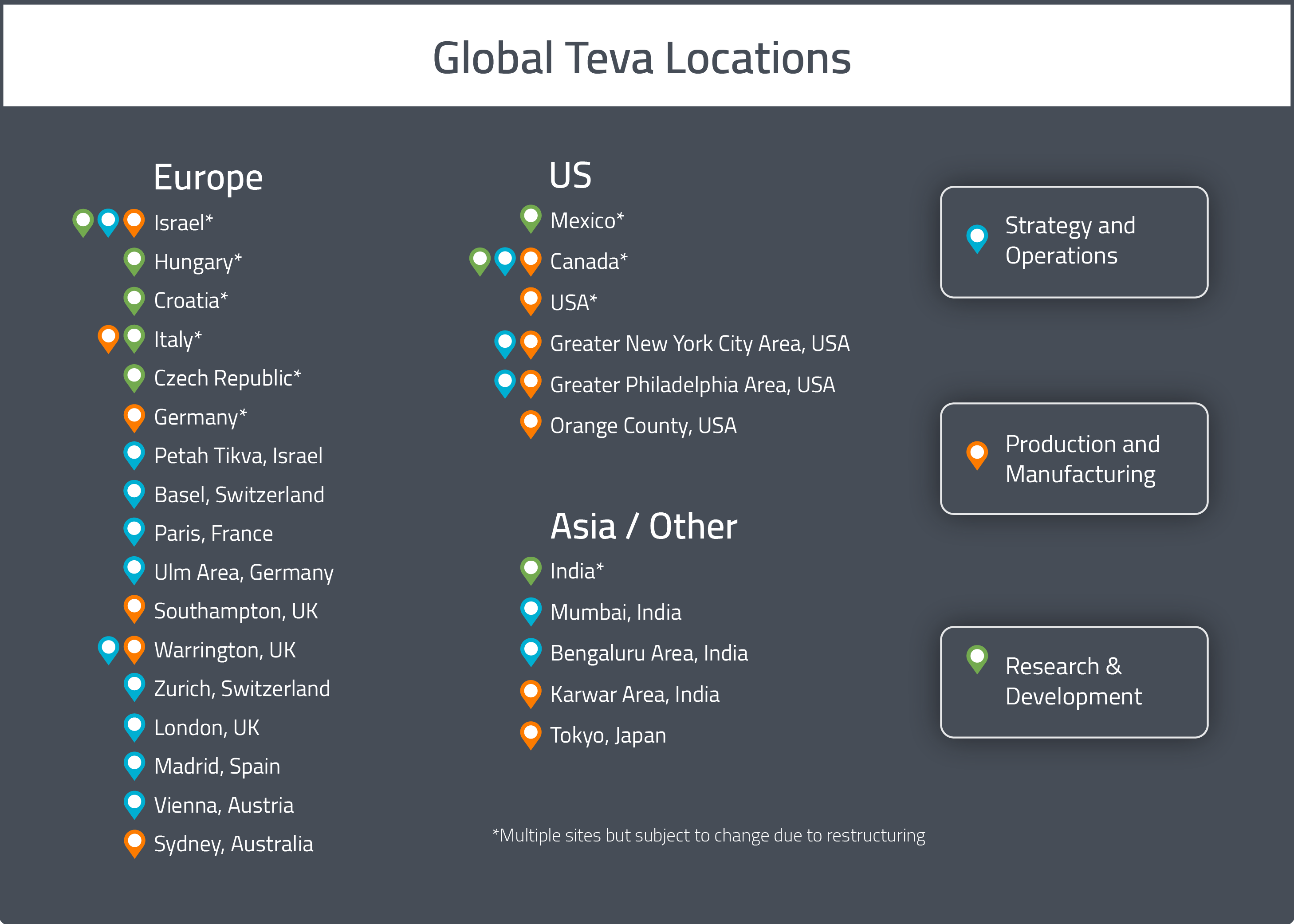
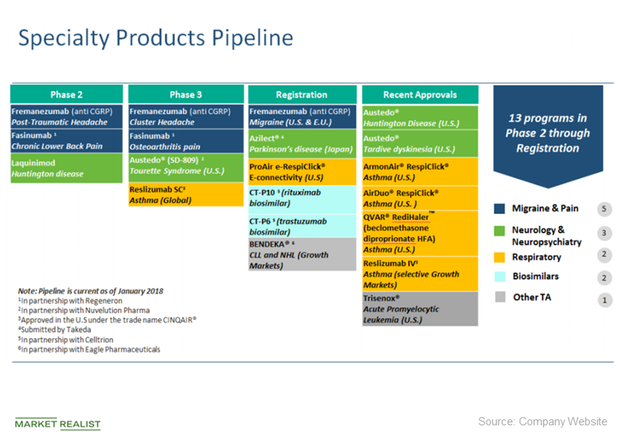
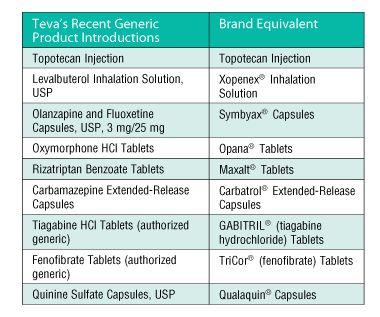
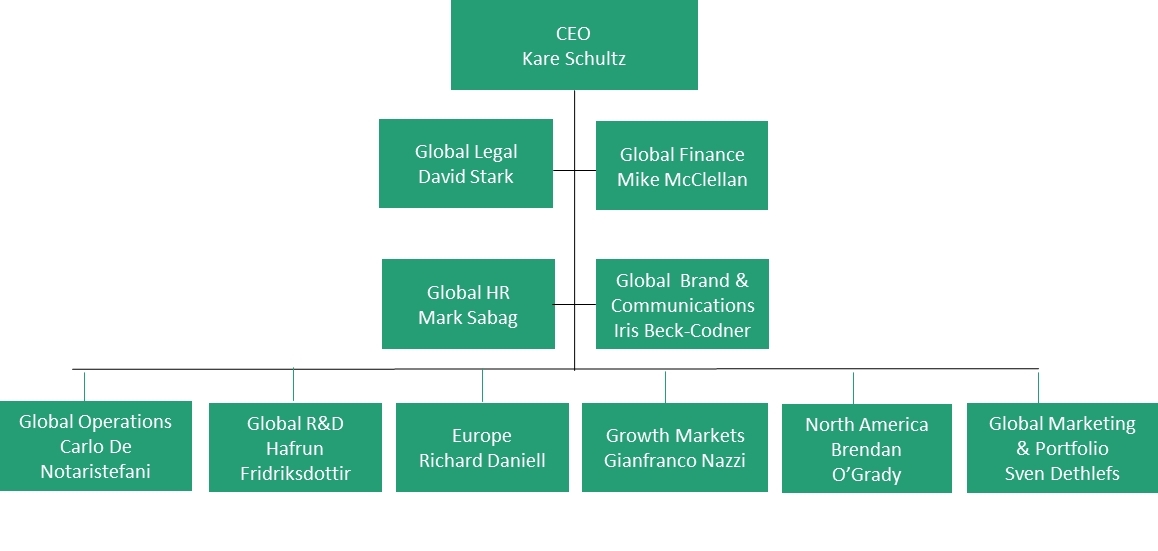
TEVA Pharmaceutical Industries Ltd. | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services
Teva, after locking up new Ajovy patents, sues Eli Lilly again in heated migraine drug fight | FiercePharma

PDF) Phase 1 dose-escalation study of the PARP inhibitor CEP-9722 as monotherapy or in combination with temozolomide in patients with solid tumors

Branded Generics Market Services Grow at a 7.3% CAGR From – Mylan N.V, Novartis AG, Teva Pharmaceutical Industries Ltd | Medgadget