New Jersey CyberKnife Develops Training Program for Multidisciplinary Approach to Lung Cancer Treatment | Toms River, NJ Patch

Adjuvant chemotherapy with or without bevacizumab in patients with resected non-small-cell lung cancer (E1505): an open-label, multicentre, randomised, phase 3 trial - The Lancet Oncology
Bristol Myers Squibb's Opdivo posts another win in early lung cancer, but debate over treatment approach likely remains | FiercePharma
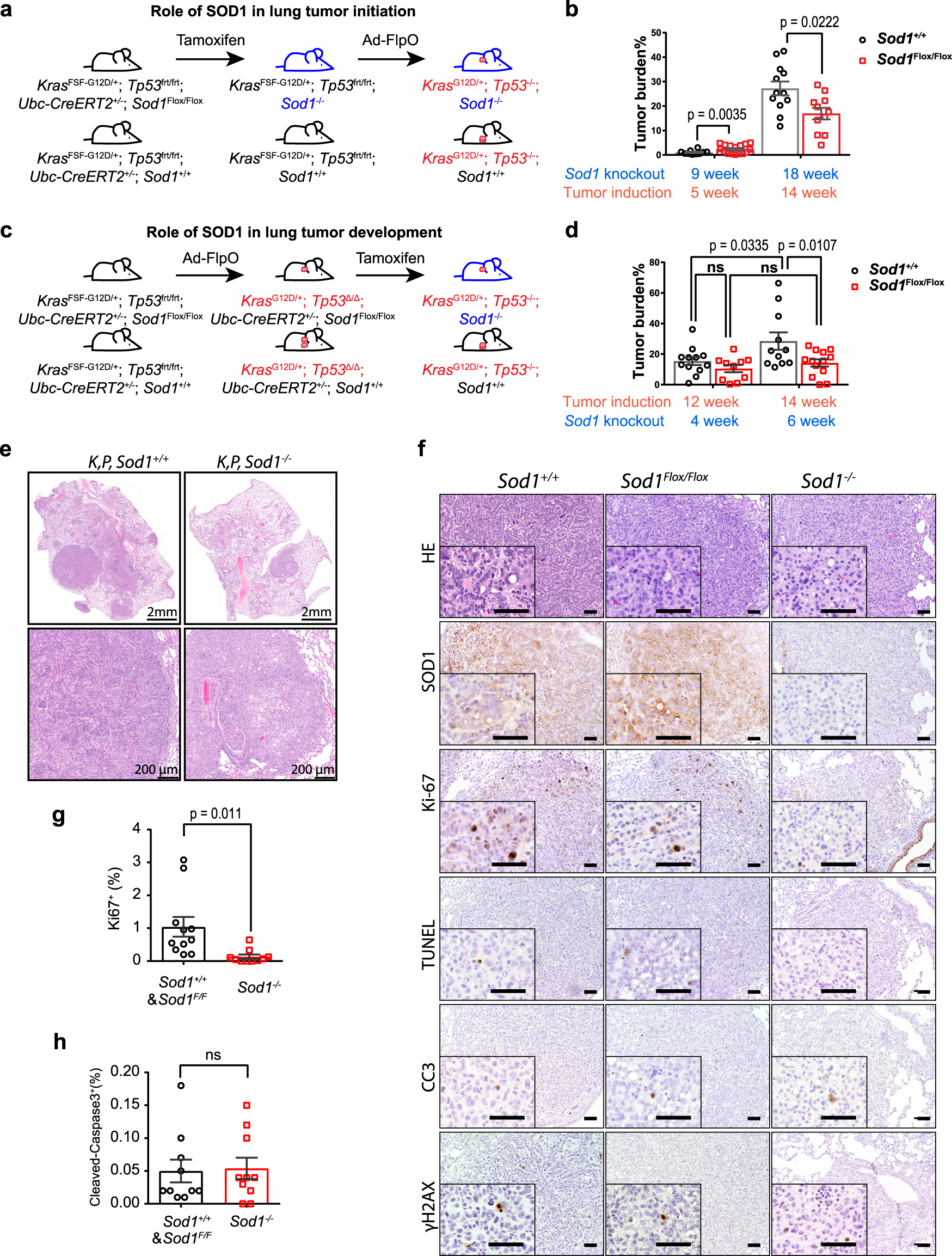
SOD1 regulates ribosome biogenesis in KRAS mutant non-small cell lung cancer | Nature Communications

First in New Jersey, and One of First in Nation, to Use New Technology for Treatment of Lung Cancer - Englewood Health

24-Month Overall Survival from KEYNOTE-021 Cohort G: Pemetrexed and Carboplatin with or without Pembrolizumab as First-Line Therapy for Advanced Nonsquamous Non–Small Cell Lung Cancer - Journal of Thoracic Oncology

Dynamic simulation of motion effects in IMAT lung SBRT – topic of research paper in Medical engineering. Download scholarly article PDF and read for free on CyberLeninka open science hub.

Durvalumab plus tremelimumab alone or in combination with low-dose or hypofractionated radiotherapy in metastatic non-small-cell lung cancer refractory to previous PD(L)-1 therapy: an open-label, multicentre, randomised, phase 2 trial - The Lancet

Lung Cancer Patient Still Going Strong a Year After Diagnosis Thanks to New Immunotherapy | Jefferson Health New Jersey

Advanced Metastatic Non-Small Cell Lung Cancer Information | ABRAXANE® (paclitaxel protein-bound particles for injectable suspension) (albumin-bound)

Cemiplimab monotherapy for first-line treatment of advanced non-small-cell lung cancer with PD-L1 of at least 50%: a multicentre, open-label, global, phase 3, randomised, controlled trial - The Lancet

Current Status and Future Perspectives on Neoadjuvant Therapy in Lung Cancer - Journal of Thoracic Oncology