First in New Jersey, and One of First in Nation, to Use New Technology for Treatment of Lung Cancer - Englewood Health
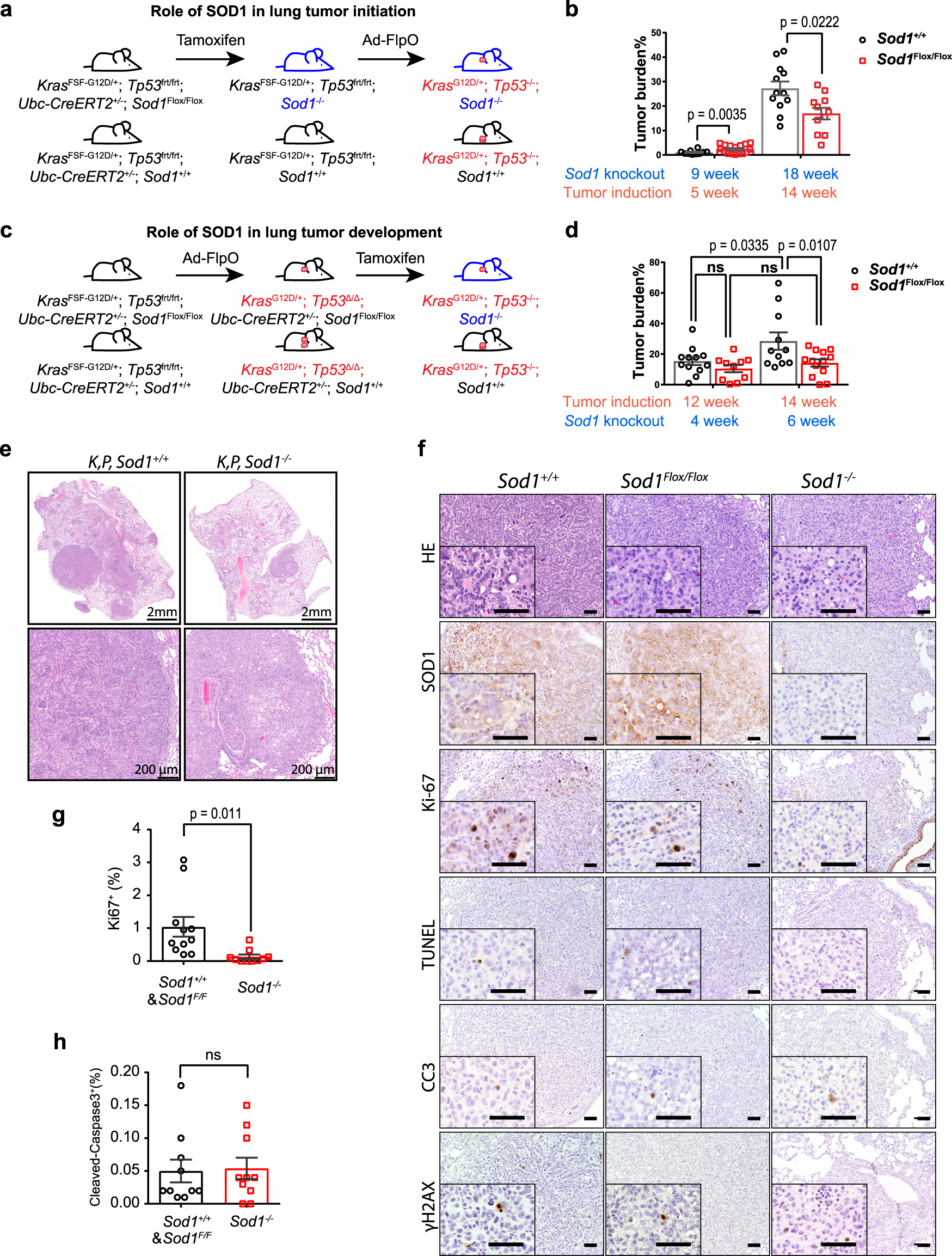
SOD1 regulates ribosome biogenesis in KRAS mutant non-small cell lung cancer | Nature Communications

24-Month Overall Survival from KEYNOTE-021 Cohort G: Pemetrexed and Carboplatin with or without Pembrolizumab as First-Line Therapy for Advanced Nonsquamous Non–Small Cell Lung Cancer - Journal of Thoracic Oncology
Systemic Therapy for Locally Advanced and Metastatic Non-Small Cell Lung Cancer | Memorial Sloan Kettering Cancer Center

Systemic Treatment Patterns With Advanced or Recurrent Non–small Cell Lung Cancer in Japan: A Retrospective Hospital Administrative Database Study - Clinical Therapeutics

Current Status and Future Perspectives on Neoadjuvant Therapy in Lung Cancer - Journal of Thoracic Oncology
Bristol Myers Squibb's Opdivo posts another win in early lung cancer, but debate over treatment approach likely remains | FiercePharma

Cemiplimab monotherapy for first-line treatment of advanced non-small-cell lung cancer with PD-L1 of at least 50%: a multicentre, open-label, global, phase 3, randomised, controlled trial - The Lancet

Advanced Metastatic Non-Small Cell Lung Cancer Information | ABRAXANE® (paclitaxel protein-bound particles for injectable suspension) (albumin-bound)